Pharma: Page 7
-
Corporate venture firms stepped in for drug startups during biotech funding pullback
According to a new BioPharma Dive analysis, corporate-related funds such as Novo Holdings, Eli Lilly and Sanofi Ventures have been among the most active investors in biotech startups this year.
By Gwendolyn Wu • Oct. 31, 2025 -
How Trump’s do-or-die agenda is impacting Big Pharma’s CEO chatter
Among tariffs, pricing policies and ongoing negotiations, pharma leaders can’t help but address their dealings with the Trump administration in third-quarter earnings reports.
By Michael Gibney • Oct. 30, 2025 -
 Explore the Trendline➔
Explore the Trendline➔
 Getty Images
Getty Images Trendline
TrendlineArtificial intelligence & machine learning
After years of excited buzz around the potential of artificial intelligence and machine learning, pharma has begun to realize the true implications and potential value of these technologies.
By PharmaVoice staff -
What comes next for Novo Nordisk?
After losing its leading position in obesity, changing its CEO and triggering a major company reorganization, the pressure is on Novo to pull off a major about-face.
By Kelly Bilodeau • Oct. 29, 2025 -
Novartis’ $12B Avidity deal shows a willingness to take big swings in neuroscience
A premium paid for RNA candidates reflects what the pharma giant has sought to do for the last few years: aim at revolutionary technology that fits into their own expertise.
By Michael Gibney • Oct. 28, 2025 -
Merck’s oncology prospects could portend a post-Keytruda future
Can the pharma giant offset potential losses from its megablockbuster with a strong suite of contenders?
By Kelly Bilodeau • Oct. 27, 2025 -
Takeda stakes more than $11B on cancer drugs from China
The Japanese pharma is licensing up to three next-generation cancer drugs from Innovent Biologics in a deal it believes to be “transformative” for its oncology portfolio.
By Ben Fidler • Oct. 24, 2025 -
Massachusetts biotech hub is limping amid layoffs and low investment
The state’s difficulties in the last year are indicative of wider industry problems during a time of market trepidation, as optimism gives way to resilience.
By Michael Gibney • Oct. 23, 2025 -
AI giants Nvidia, Microsoft and Google are making critical moves in pharma R&D
With market caps in the trillions, these tech behemoths are establishing a growing presence in drug development. Here’s what they’re up to.
By Alexandra Pecci • Oct. 22, 2025 -
AstraZeneca, Daiichi’s Datroway excels in hard-to-treat breast cancer
Presented at the European Society for Medical Oncology, study results showed Datroway extended survival in breast cancer patients for whom immunotherapy is not an option.
By Delilah Alvarado • Oct. 21, 2025 -
In the Salesforce vs. Veeva saga, the clock is ticking for drugmakers
The CRM platform split will be official in 2030, but that still doesn’t leave much time for pharmas and biotechs that have yet to choose which way to go.
By Michael Gibney • Oct. 21, 2025 -
Why a treatment older than the FDA is getting new regulatory scrutiny
MAHA-aligned patients and providers are pushing back to ensure access to an animal-derived thyroid hormone treatment.
By Kelly Bilodeau • Oct. 20, 2025 -
Pregnant women, long excluded from drug trials, are back in the spotlight
The White House has raised concerns around women’s health, but R&D on pregnant and breastfeeding women is still lacking.
By Meagan Parrish • Oct. 17, 2025 -
J&J’s orthopedics selloff keeps all eyes on a thriving pharma portfolio
As another J&J department bites the dust, leadership is focused on what’s working in the pharma business, and there’s plenty to see.
By Michael Gibney • Oct. 16, 2025 -
Too much of a good thing? A flood of data is slowing clinical trials.
Pharma companies want to know it all — but cutting out unnecessary data collection could speed drug R&D.
By Kelly Bilodeau • Oct. 15, 2025 -
Ultra-rare diseases got a boost from recent FDA approval signalling more regulatory flexibility
Stealth BioTherapeutics’ FDA win could help build momentum in the broader rare disease space.
By Alexandra Pecci • Oct. 15, 2025 -
What a gene therapy win means for the Huntington’s disease pipeline
Recent results for a Huntington’s gene therapy confer a hopeful sign for R&D in the space.
By Kelly Bilodeau • Oct. 14, 2025 -
Beyond dire wolves: Could Colossal’s de-extinction work transform human health?
While resurrected animal species grab headlines, Colossal’s behind-the-scenes R&D may be poised to make an impact for humans as well.
By Meagan Parrish • Oct. 10, 2025 -
Nobel Prize victory spotlights immunology’s value in pharma
While saturated with mature blockbusters, the immunology space is vibrating with new potential breadwinners from the top companies.
By Michael Gibney • Oct. 9, 2025 -

 Ermath, Michael. (2020). "Individualized Therapies Workshop" [Photograph]. Retrieved from Flickr.
Ermath, Michael. (2020). "Individualized Therapies Workshop" [Photograph]. Retrieved from Flickr.
Dr. Peter Marks, former top FDA vaccine official, joins Eli Lilly
Six months after his abrupt resignation as CBER director, Marks has been hired to run discovery and infectious disease work at the big Indianapolis drugmaker.
By Jonathan Gardner • Oct. 8, 2025 -
Vertex’s quest for a breakthrough in Type 1 diabetes
The race is on for an insulin-producing treatment that doesn’t require immune suppressing drugs.
By Kelly Bilodeau • Oct. 8, 2025 -
Q&A
Overcoming past mistakes, generics powerhouse Teva embraces an innovative ‘mindset change’
In the three years since a leadership change, Teva has evolved its R&D approach beyond a long history making copycat medicines.
By Michael Gibney • Oct. 7, 2025 -
Trump administration could lower drug prices by making other nations pay more
The Trump administration often touts its efforts to lower prices in the U.S. by matching what other countries pay, but there’s more to this policy story.
By Kelly Bilodeau • Oct. 6, 2025 -
With Emma Walmsley exiting GSK, who will be pharma’s top women CEOs?
The only woman to head up a company in Big Pharma is stepping down from the role at the end of the year.
By Meagan Parrish • Oct. 3, 2025 -
Takeda, in reversal, abandons cell therapy research
The Japanese pharmaceutical company, which had made cell therapy a priority a few years ago, aims to partner its work while prioritizing investments elsewhere.
By Delilah Alvarado • Oct. 2, 2025 -
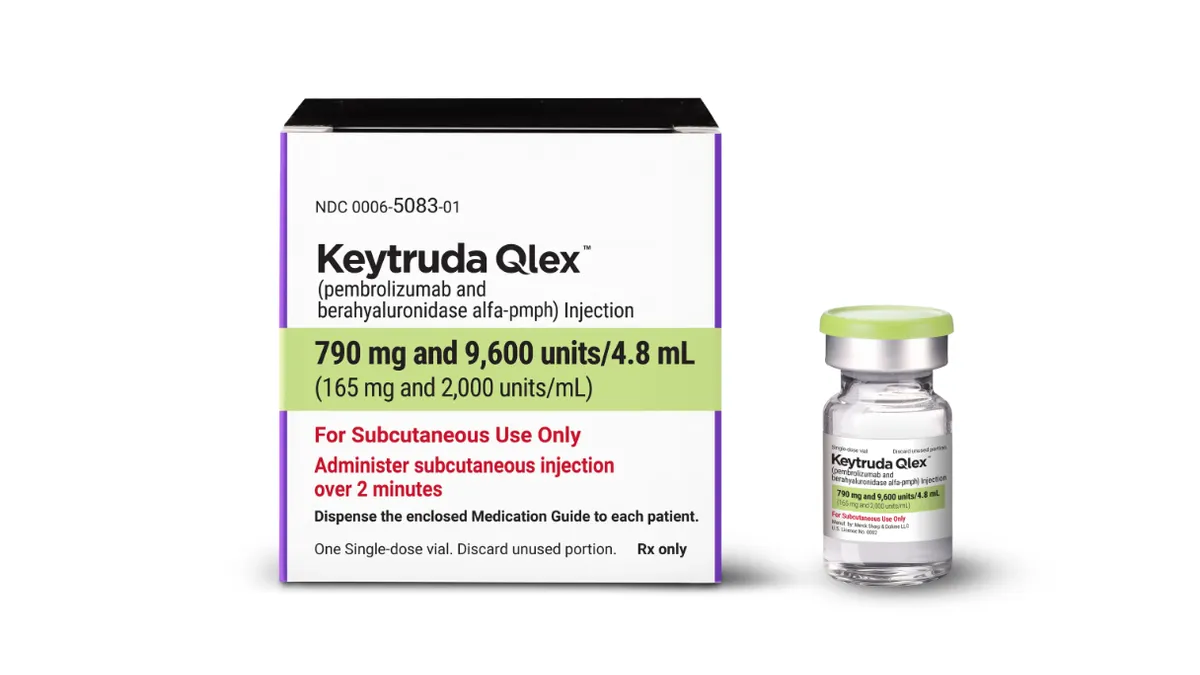
Half of Merck’s sales are in jeopardy. Can Keytruda’s sequel save the day?
This week’s launch of subcutaneously administered Keytruda Qlex gives Merck a safety net for the cancer drug’s daunting patent cliff.
By Michael Gibney • Oct. 2, 2025